
Nail Composition
Nails are made up of layers of a protein called “keratin,” which grows naturally from the base of nail under the cuticle area. Like all proteins, keratin is made up of building blocks called “amino acids,” which are linked together in chains to make a functional structure. Bonds between chains (crosslinks) play an important role in making keratin strong and resilient. Any damage to these amino acid chains or crosslinks will weaken the nail.
Nail Damage
Nails are damaged by normal wear-and-tear in everyday activities, ranging from typing to gardening to chores. Most nail damage is also attributed to household chemicals including soap, cleansers and hand sanitizers. As most nail professionals are sadly aware, a major cause of nail damage is improper manicure or pedicure procedures at that nonstandard salon down the street—which you, as a good nail tech, are expected to magically fix! 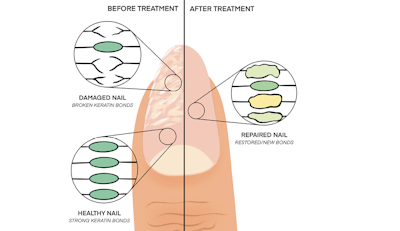 Photo provided by Sarah Ostresh
Photo provided by Sarah Ostresh
Keratin bonds can be broken by aggressively buffing the nails, misuse of drills, over-filing or impatiently peeling off enhancements–all of which leave the nails thin, weak, fragile and, in some cases, with peeling layers or grooves (“rings of fire”) from excess drilling.
Another source of nail damage is the overuse of hardening products. This creates the opposite problem; too many crosslinks leave the nail too stiff and prone to splitting or peeling. Additionally, nail damage can be caused by allergies to nail products, fungal infections and the misguided practice of cuticle removal, all of which can cause inflammation of the surrounding tissue.
Nail Treatment
When a client comes in with devastated nails, where do you begin? Fortunately, nail treatments can dramatically improve the condition of nails. They are classified under three types based on their ingredients and mechanism: nourishing nail and cuticle treatments, firm-forming treatments and crosslinking treatments.
Nourishing nail and cuticle treatments are formulated with hydrating and nourishing ingredients such as grapeseed, sesame, kukui, sunflower or avocado oils. These products replenish the natural oil content of the skin, cuticles and nails. Badly damaged nails should be treated this way for a while, to give them a chance to recover.
Film forming treatments are modifications of nail lacquers or gels, either clear or colored. The product works by forming a hard film, which provides a protective coating on the nail surface. Therefore, the benefits come from the added strength and durability of the coating itself rather than altering the physical properties of the nail. By imparting a hard and glossy film, the product reinforces the nail, preventing it from breaking. A note of historical interest, this is how the modern nail industry began–In 1957, a dentist strengthened his broken fingernail with dental acrylic.
Crosslinking treatments alter the nail protein by building bonds between keratin strands. Historically, formaldehyde was the standard nail hardener, as it is an effective crosslinker for keratin. However, despite the almost unmeasurably low systemic formaldehyde exposure during hardener use, formaldehyde was banned for nail hardeners in Japan and the EU, and other jurisdictions are considering similar rules. Therefore, the industry has explored alternate crosslinkers such as glyoxal, dimethyl urea, hexanal and others. A newer technology, recently patented by OPI, relies on biomimicry of the keratin structure, building new bonds from within to strengthen the nail and repairing the keratin by up to 99%.
Healthy Nails Going Forward
As Heather Reynosa, director of global education design for OPI, puts it, “You need to be interested in the client’s lifestyle and holistic care of the nail.” Understanding how clients stress their nails will give you guidance as to what type of product to select and ongoing service to provide.
Sunan Yuvavanich, M.S., is a principal scientist with OPI.
Keratin Repair Research
By: Laurisa London-Dawodu, Ph.D., Principal Analytical Scientist, OPI
Hair and nails are made up of similar structural fibrous keratin proteins. Over time, due to the exposure to various physiochemical forces, the secondary protein structure of keratins can be damaged by the disruption of the various protein cross-linkages found in keratin, such as ionic, disulfide and hydrogen bonds. Moreover, water affects the keratin structure in many ways, through the breaking of ionic bonds and the replacement with weaker hydrogen bonds. Mitigation efforts have been made in the repair of damage using various treatments. Dual approaches have been employed in the repair of keratin substrates, targeting of the internal structure using bond-builders and applying high molecular weight film formers on the outer surfaces. While both pathways aim to ameliorate damage by increasing the break stress and preventing premature fractures, the focus will be on internal repair.
In the realm of hair care, engineered smart materials penetrate and target the internal keratin structure using several mechanisms of repair. One path is in-situ polymerization of maleic acid during the high-lifting coloring process in the presence of persulphates and polyvinylpyrrolidone (PVP). Another repair mechanism is covalent cross-linking, whereby rebuilding of the broken disulfide bonds is accomplished using a chemical bridge. This is believed to be accomplished through the reaction of maleic acid with the thiol groups resulting from broken disulfide linkages. Thirdly, protein and peptides have been thoroughly researched for their ability to penetrate and bind to keratin, resulting in improved straightening and strengthening properties.
















